SOFTWARE
Assay setup, data analysis, and report generation software built for your scientific achievement
Our Software
Built for a touch screen with easy-to-follow, walk-through steps, Gator® software provides users with assay setup, data analysis and report generation in one complete, integrated software package.
Our software, GatorOne Software, guarantees complete flexibility, making it possible for the user to conduct experiments across a wide spectrum — from the routine to the more complicated and novel. The same holds true for data analysis, where you can create templates your entire team can use to ensure the analysis is done systematically across many users — or to make sure each assay is analyzed uniquely.
GatorOne v2.17 – Released April 2025
Gator® Navigator Software
Fast data processing, easy visualization and reporting speeds up your biotherapeutics discovery. Multiple assays can be combined into a larger binning matrix. By combining eight 32 x 32 binning assays, the analysis for one 96 x 96 competition profile can be achieved in less than 5 minutes.
Gator® Part11 Software – 21 CFR Part 11 Compliant
The FDA’s code of Federal Regulations (CFR) Title 21 is critical in ensuring safe and ethical drug administration. Gator® Part11 Software enables users in GMP or GLP laboratories environment to comply with 21 CFR Part 11 regulations. All data acquired is time-stamped and traceable. Features such as account management, enhanced audit trails, and recorded user sessions comply with FDA guidance.
Continuously striving for better assay setup
Quantitation, Kinetics, and Regeneration in one run
These modes enable users to start assays faster.
More than just a software, Gator® uses predictive logic to generate assay maps for common experiments, such as epitope binning. Additionally, we use built-in AI logic to determine whether assays are set up incorrectly, offering solutions before mistakes occur. Once an assay protocol is generated, it can be saved and stored in a user’s personal folder, enabling future assay starts in just two clicks.

Designed to bring you the best data analysis
Each Gator® quick-start mode has data analysis templates prebuilt, making analysis a breeze. The software contains a robust set of fitting models for standard curve analysis and fittings, as well as for kinetic applications. Built-in logic can help users detect whether a particular fitting model is less-than ideal for helping new or experienced users conduct experiments. What’s more, in just two clicks, epitope binning data analysis can generate a traffic light pattern of results and a matrix of interactions to provide a rich data set.
Focused on figure and report generation

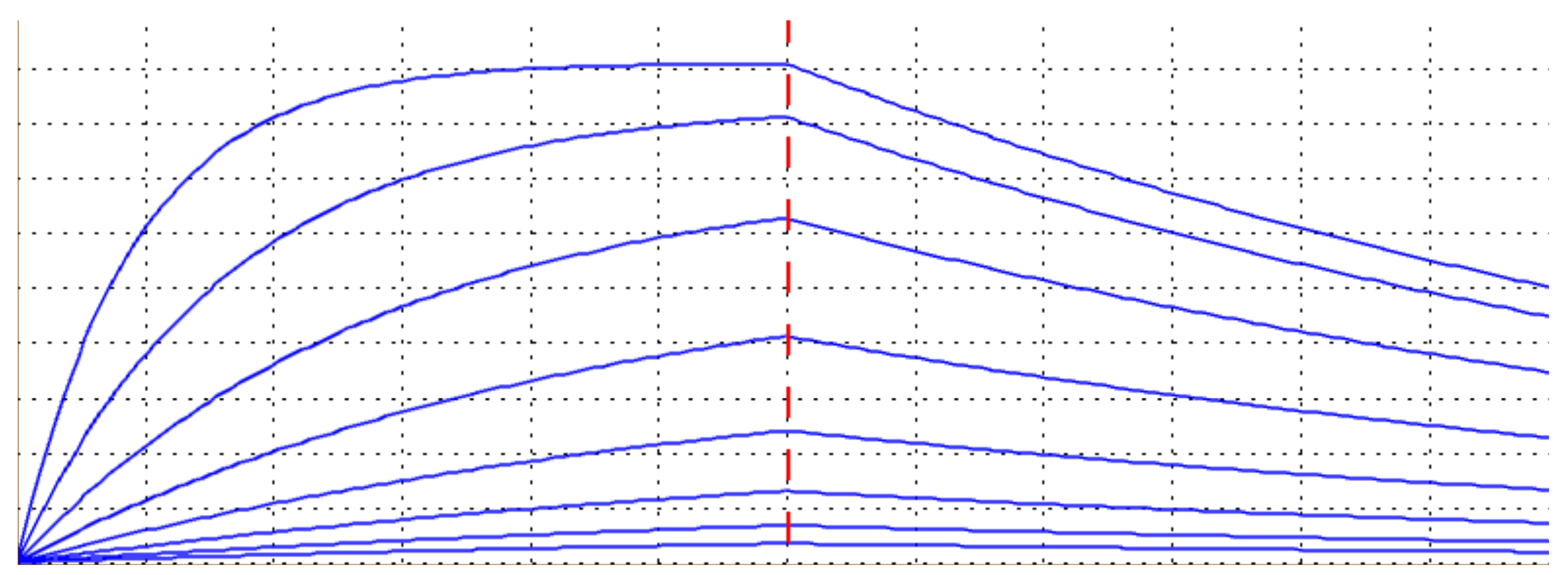
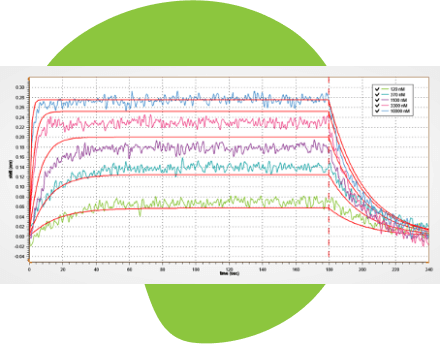
A Sensogram Simulator for Kinetics Assays
Our software has a unique sensogram simulator for generating the optimal association and dissociation curves for a given analyte titration. It saves time and resources, while enhancing confidence in the experimental design and data.
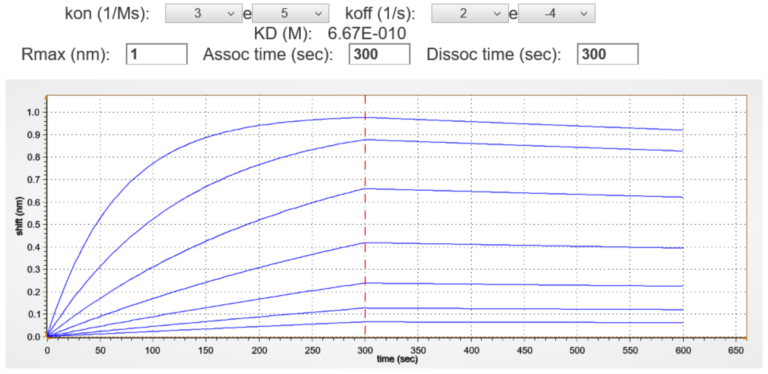
Concentration Series Too High
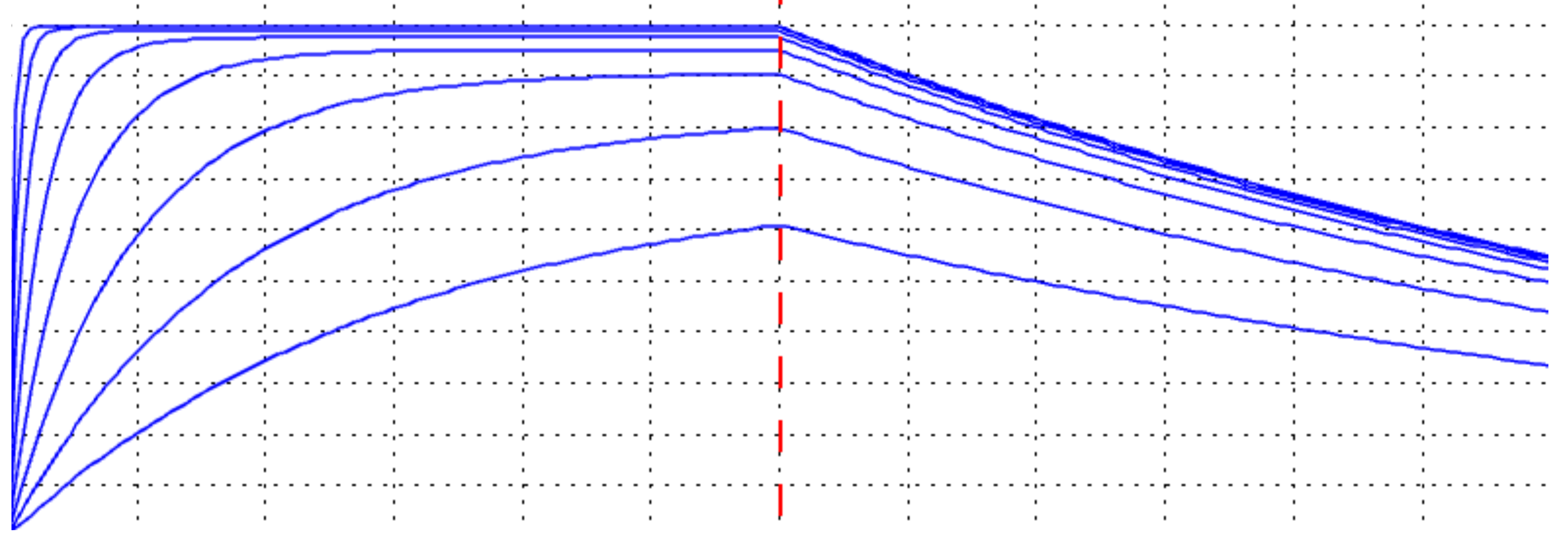
Concentration Series Too Low
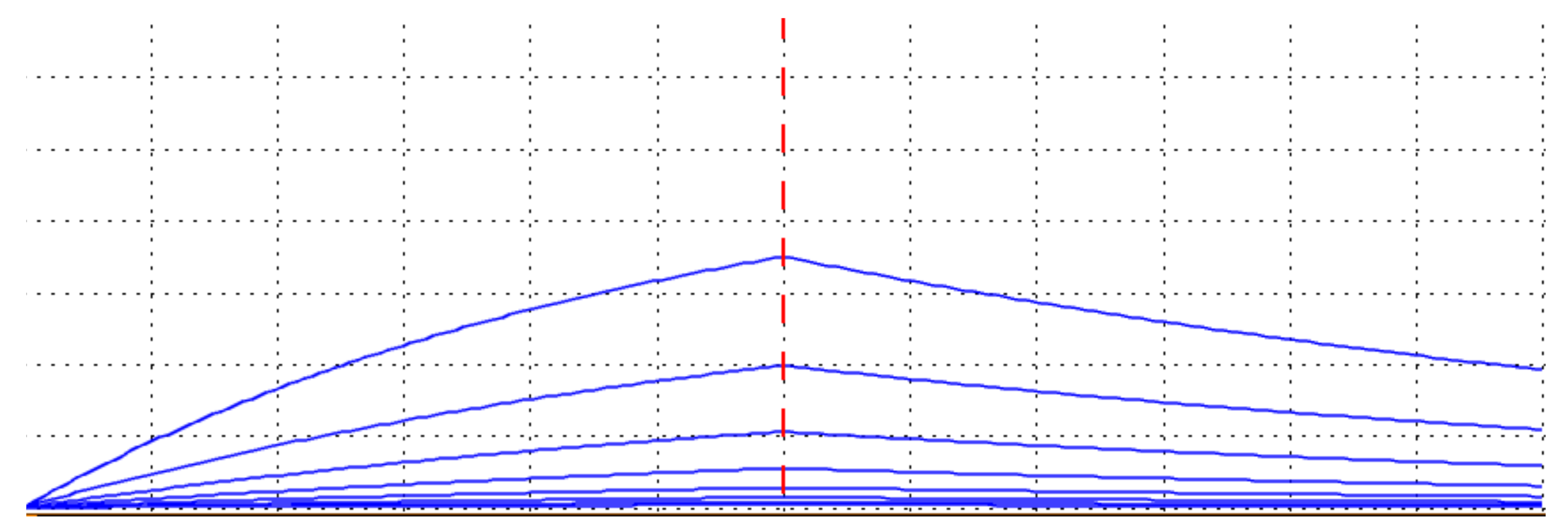
Optimal Concentration Series